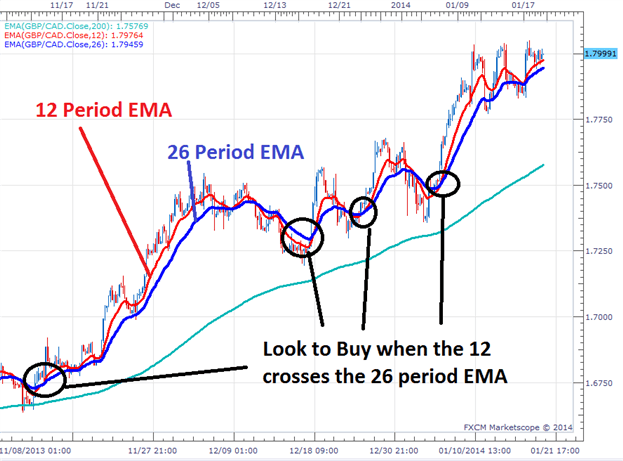
Must be CE marked in accordance with EU medical devices legislationĮMA is responsible for evaluating the quality, safety and efficacy of marketing authorisation applications assessed through the centralised procedure, including the safety and performance of a medical device in relation to its use with a medicinal product.tablet delivery system with controller for pain management.Medicinal product and device are separate items contained in same pack or obtained separately.Requirement does not apply to Class I devices (non-sterile, non-measuring).Requirement applies since under article 117 of Medical Devices Regulation.Marketing authorisation application should include a CE certificate for the device or, if not CE marked but would need to be certified if marketed separately, applicant must include an opinion from a notified body on conformity of device.Medicinal product and device form a single integrated product.The device part of the combination may require a conformity assessment, as follows: Type of combination The entire product is regulated under EU pharmaceutical legislation ( Directive 2001/83/EC or Regulation (EC) No 726/2004) and must obtain a marketing authorisation for a medicinal product. If the principal intended action is achieved by the medicine, it is considered a medicinal product that includes a medical device. Some medicines are used in combination with a medical device, usually to enable the delivery of the medicine. High-risk medical devices ─ EMA supports the medical device expert panels that provide opinions and views to notified bodies on the scientific assessment of certain high-risk medical devices and in vitro diagnostics.The EMA provides scientific opinions on the compliance of the substance with the requirements laid down in Annex I to Directive 2001/83/EC. 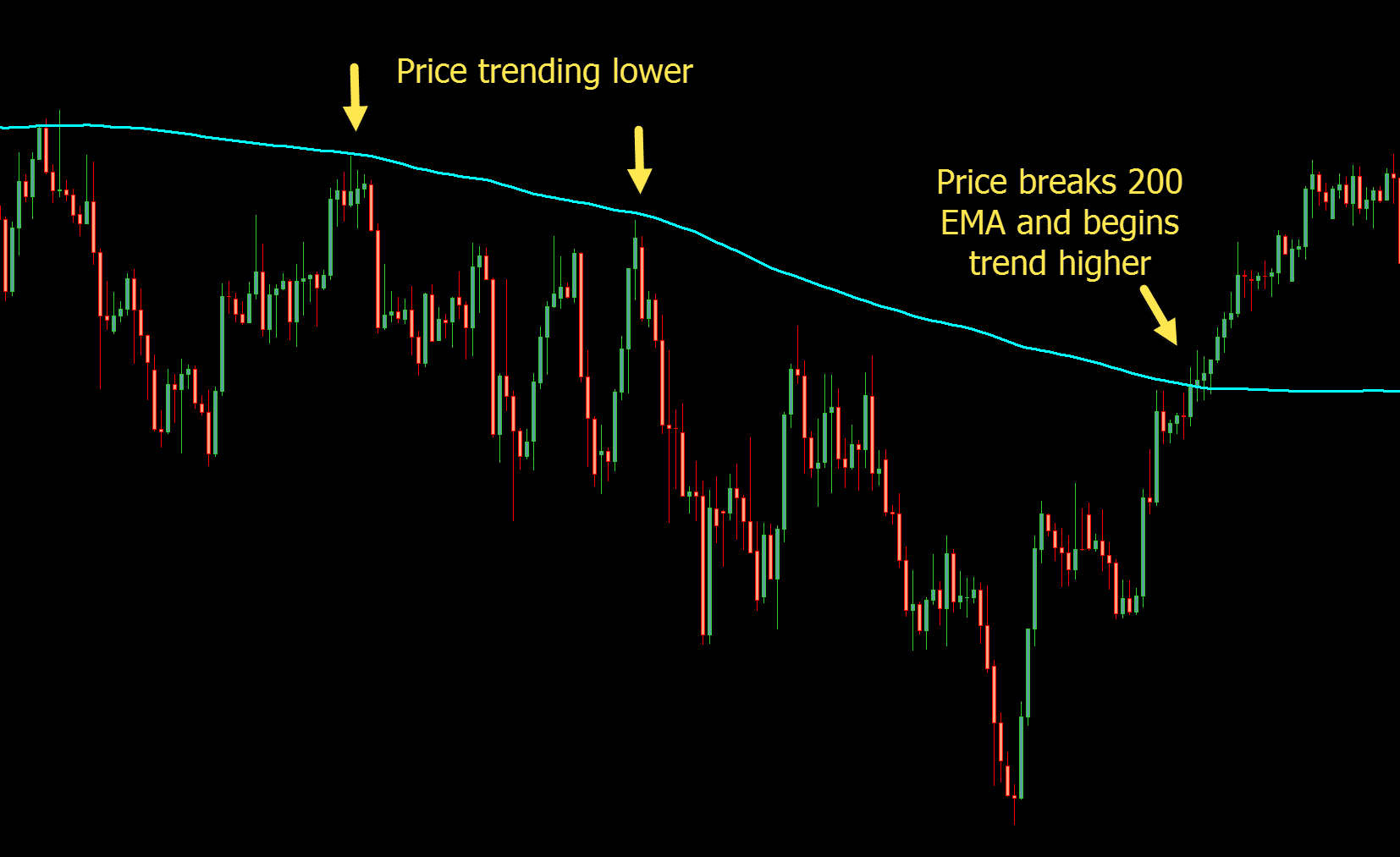
Medical devices made of substances that are systemically absorbed ─ the notified body must seek the scientific opinion of a competent authority.Companion diagnostics ('in vitro diagnostics') ─ the notified body must seek EMA's scientific opinion on the suitability of the companion diagnostic to the medicinal product if the latter falls within the scope of the centralised procedure.Medical devices with an ancillary medicinal substance ─ the notified body must seek EMA's scientific opinion on the quality, safety, and usefulness of the ancillary medicinal substance in three cases: if the ancillary substance is derived from human blood or plasma if it has been previously evaluated by the EMA or if it falls within the mandatory scope of the centralised procedure.This is part of a centralised procedure application for the medicinal product. Medicines used in combination with a medical device ─ EMA assesses the safety and effectiveness of medicines used in combination with a medical device. 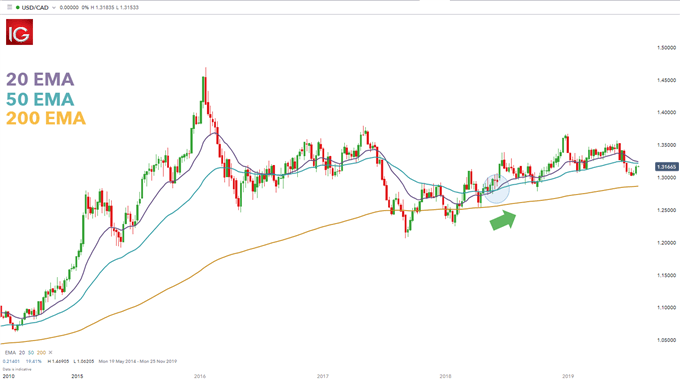
In some other cases, the notified body must seek a scientific opinion from EMA before issuing a CE certificate.ĮMA has distinct regulatory responsibilities per category of medical device, including in vitro diagnostics. These expert panels benefit from EMA's technical and scientific support. The conformity assessment usually involves an audit of the manufacturer's quality system and, depending on the type of device, a review of technical documentation from the manufacturer on the safety and performance of the device.ĮU Member States designate accredited notified bodies to conduct conformity assessments. For certain high-risk devices, notified bodies shall request the opinion of specific expert panels before issuing the certificate of conformity. Manufacturers can place a CE (Conformité Européenne) mark on a medical device once it has passed a conformity assessment. They are regulated at EU Member State level, but the European Medicines Agency (EMA) is involved in the regulatory process. 
In the European Union (EU) they must undergo a conformity assessment to demonstrate they meet legal requirements to ensure they are safe and perform as intended. Medical devices are products or equipment intended for a medical purpose.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
